AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Mesenchymal stem cells12/13/2022   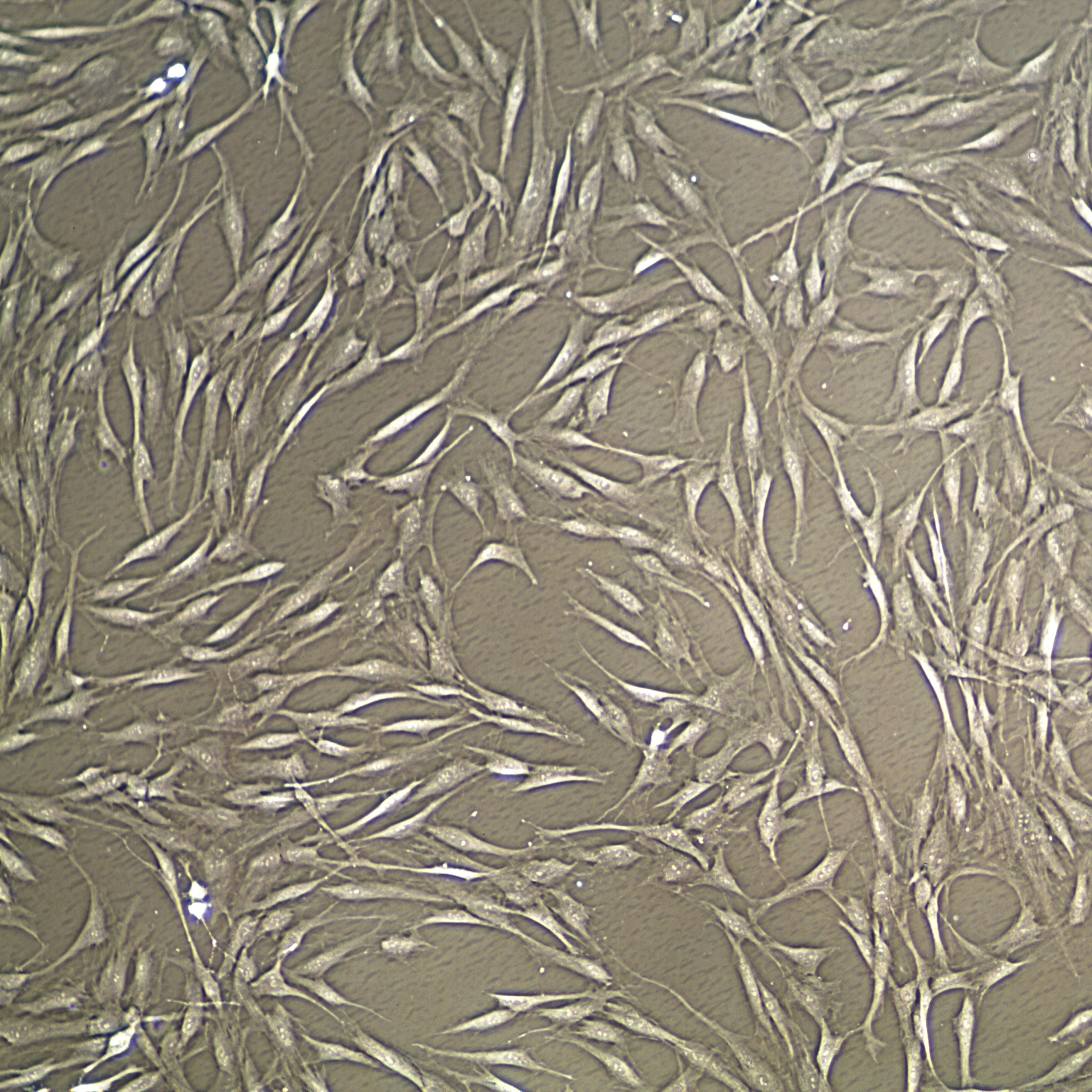
in Physical Biochemistry from Newcastle University ( U.K.) and has authored more than 100 papers and abstracts many of which are in the cell and gene therapy space. Craig brings to jCyte over three decades of experience in the implementation of technical operations, quality systems, and regulatory affairs for complex biologics, including hematopoietic stem cells (HSCs), mesenchymal stem cells (MSCs), neural stem cells (NSCs), activated T cells, gene-modified T cells and gene-modified HSC products, as well as recombinant proteins, monoclonal antibodies, adeno-associated virus (AAV), gamma-retroviral (gRV), and lentiviral (LV) vector products. Craig will be the strategic and technical lead for all drug product development activities, including manufacturing process development, GMP facility design, build-out and scale-up, technology transfer, analytical test method development, quality control, drug product formulation development, and CMC regulatory interactions and filings.Īn accomplished biotechnology executive and scientist, Dr. Stewart Craig as Chief Technology Officer. jCyte, Inc., a private biotechnology company dedicated to improving the lives of patients with retinal degenerative diseases, announced the expansion of the company’s leadership team with the appointment of Dr. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
